

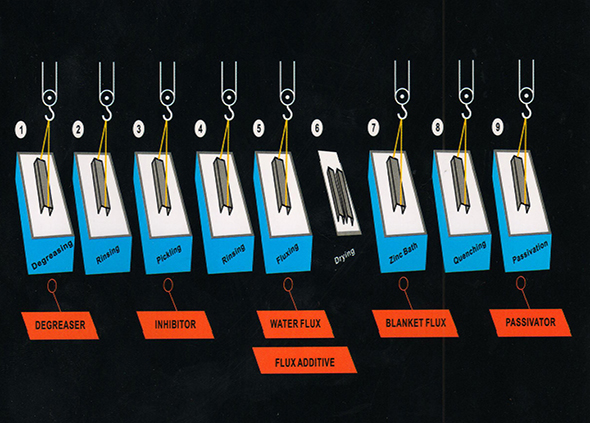
It is a process which has been carried out for more than 300 years now, The exercise of coating Steel with Zinc is one of the best options for protecting the metal. The metal is immersed in a bath of molten zinc at a temperature of 455 C (840 F), which alloys the surface of the base metal. This practice still remains the most popular and in demand.

Our acidic degreaser can be used with water and hydrochloric acid to support two actions at the same time; degreasing and pickling. The degreaser also provides an excellent base for further operations of galvanizing. Our degreasers not only remove the organic contaminants (dirt, grease, oil, wax, grime and dirt), but also accelerate the derusting of the surface of the metal.
This is a heavy duty water based alkaline, which is also a self generating cleaner. It converts approximately 30-40% of waste fatty contents (vegetable oil and fatty acids) into an active cleaner, or a soap.
Passivation is a chemical process of treating the metal to form an oxide bond that enhances the original corrosion resistance by forming a thin transparent oxide coating. Passivator provides maximum protection from corrosion on newly hot dipped galvanized steel.
The passivator contains a special additive which acts as a water conditioner and controls the concentration of zinc and iron in the bath, thereby elongating the life of the bath nearly twice as much as any other process. It comprises chrome third and sixth salt composition to seal and passivate the galvanize surface.
This passivator is free from chrome, and is made by mixing polymers and phosphates to seal and passivate the galvanized surface. This low strength solution forms a passive film on a fresh metal surface, and prevents further oxidation and tarnishing.
Our inhibitors provide a bright and a smooth surface. The accumulation of our
inhibitors ensures less pollution, acid consumption and undesired metal loss. It also decreases the rate
of metal oxidation. Corrosion inhibitors minimize the undue attack of acid, on the surface of steel in a
pickling solution.
The inhibitor is available for hydrochloric acid (HCL) and sulphuric acid (H2SO4)